Herpes Infection Treatment Market : Global Share, Size, Growth, Trends & Outlook ( 2022 – 2032 )
Herpes Infection & Treatment Insights:
The Herpes simplex virus, or HSV, is the cause of the viral infection known as Herpes. Herpes simplex virus comes in two forms: HSVs I and II. The most common form of oral Herpes caused by HSV-1 is cold sores or fever blisters on the face or around the mouth. Genital Herpes, characterized by painful sores or blisters on or around the rectum or genitals, is typically caused by HSV-2. Herpes is a virus that is extremely contagious and can be spread through sexual contact, kissing, or sharing personal items like towels or razors.
The presence of symptoms, such as cold sores or genital sores, and laboratory testing, such as a viral culture or PCR test, are typically used to diagnose Herpes infections. Herpes infections are typically treated with antiviral medications like acyclovir, valacyclovir, and famciclovir as the mainstay. These medications prevent the virus from reproducing, thereby reducing symptoms' duration and severity. Over-the-counter pain relievers and topical creams, in addition to antiviral medications, can be used to ease Herpes outbreak-related pain and discomfort. There is currently no vaccine for the Herpes simplex virus, but some vaccines are in development and some clinical trials are ongoing.
Herpes Infection Treatment Emerging drugs
Several new drugs are currently in development for the treatment of Herpes infections. Some examples include:
- Pritelivir: This antiviral medication lessens the likelihood that the Herpes simplex virus will develop resistance because it targets a different stage of the virus's replication than other medications do. In clinical trials, Pritelivir has produced encouraging outcomes, and it is currently undergoing Phase III clinical trials.
- Brivudin: Brivudin similar to acyclovir and valacyclovir, has a longer half-life, which means it stays active in the body for a longer duration. Brivudin is being studied for the treatment of other Herpes infections and has been approved for the treatment of Herpes zoster (shingles) in some countries.
- Maribavir: This antiviral medication lessens the likelihood that the Herpes simplex virus will develop resistance because it targets a different stage of the virus's replication than other medications do. Maribavir is currently undergoing Phase III clinical trials, and its clinical trials have shown promising results.
- Brincidofovir: Multiple DNA viruses, including cytomegalovirus and adenovirus, have been targeted and treated by Brincidofovir antiviral medication. The Herpes simplex virus treatment by this medication is positive in Phase III clinical trials.
Herpes Infection Treatment Market Size and Share
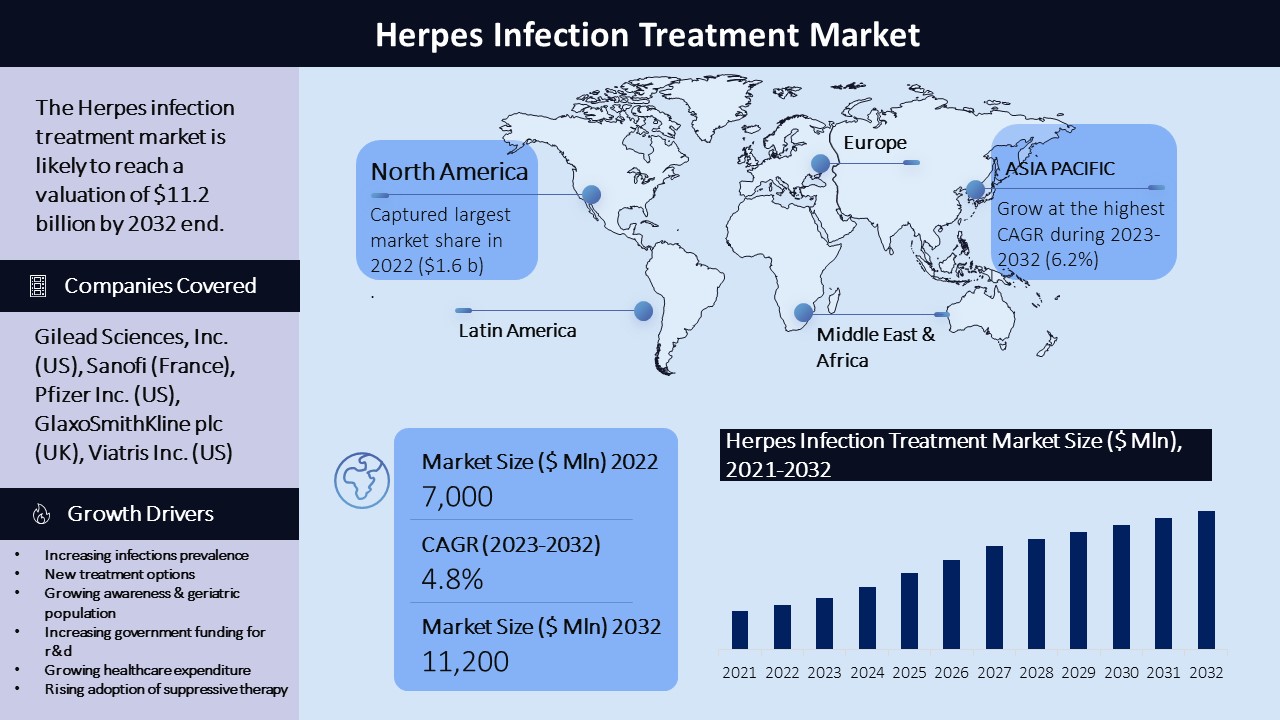
The Herpes infection treatment market's compound annual growth rate (CAGR) value is expected to fluctuate based on factors like the prevalence of the Herpes simplex virus and the availability of treatment options. The Herpes Infection Treatment Market was valued at approximately $7.0 billion in 2022 and is anticipated to generate approximately $11.2 billion in revenue by the end of 2028, expanding at a CAGR of approximately 4.8% between the years 2023 and 2032.
A subsegment of the pharmaceutical industry, the Herpes infection treatment market focuses on the creation and sale of medications to treat Herpes infections. Antiviral medications like acyclovir, valacyclovir, and famciclovir are common treatments because they stop the virus from reproducing. In clinical trials, some more recent medications, like pritelivir, have demonstrated promising results.
Herpes Infection Treatment Market Driving Factors:
Several factors are driving the growth of the Herpes infection treatment market including:
- Increasing prevalence of Herpes infections: More than 400 million people between the ages of 15 and 49 have Herpes simplex virus type 2 (HSV-2) and more than 3.7 billion people under the age of 50 have oral Herpes, according to the World Health Organization (WHO).
- Development of new treatment options: The market for Herpes infection treatment is anticipated to expand driven by the development of new medications like pritelivir and brivudin and ongoing vaccine research.
- Growing awareness & geriatric population: The number of elderly people is increasing worldwide, and are more likely to develop Herpes. With the increasing awareness about Herpes, more people are seeking treatment for their symptoms.
- Increase in government funding for research and development: It is anticipated that government funding for research and development will rise, driving the event of the latest Herpes vaccines and medicines.
- Growing healthcare expenditure: Rising healthcare costs, both public and private, are driving investment in research and development of new treatments for the market for Herpes infection treatment.
- Rising adoption of suppressive therapy: The use of suppressive therapy is also rising in tandem with the rise in Herpes recurrence. The market for Herpes infection treatment is anticipated to expand in response to this trend.
Herpes Infection Treatment Market Key Trend & Developments:
There have been several recent developments in the Herpes infection treatment industry, including:
- Approval of new drugs: Valtrex, a topical cream for the treatment of recurrent oral Herpes (cold sores) caused by Herpes simplex virus type 1 (HSV-1) was approved by the FDA in 2021.
- Collaborations and partnerships: New drugs, vaccines, and diagnostic tools are being developed in collaboration with other businesses, research institutions, and government agencies by several Herpes infection treatment companies.
- Increase in awareness campaigns: Herpes awareness campaigns are on the increase, which is predicted to scale back the stigma related to the disease and increase the diagnosis and treatment rates.
- Increase in mergers and acquisitions: The Herpes infection treatment industry has seen several recent mergers and acquisitions, such as Mylan's 2019 merger with Upjohn, a Pfizer division, and Mylan's 2016 acquisition of Meda AB. Market consolidations could result in more cost-effective and efficient treatment options.
- Increase in R&D investment: As companies focus on developing new Herpes drugs and vaccines, the Herpes treatment market has seen an increase in R&D investment.
- New drugs in development: For the treatment of Herpes infections, several new medications including pritelivir, brivudin, maribavir, and brincidofovir, are currently in development which could be simpler and have fewer side effects compared to the present treatment options.
Herpes Infection Treatment Market Segmentation:
The Herpes infection treatment market can be segmented based on the following:
- Type of Herpes virus: Herpes simplex virus type 1 (HSV-1), Herpes simplex virus type 2 (HSV-2), Varicella-zoster virus (VZV)
- Distribution channel: Hospital pharmacies, Retail pharmacies, e-commerce
- End-user: Hospitals, Clinics, Homecare
- Route of Administration: Oral, Topical, Injectable
- Treatment type: Antiviral drugs, Topical treatments, Immunotherapy
- Geography: North America, Europe, Asia Pacific, Latin America, Middle East & Africa
Herpes Infection Treatment Market Regional Synopsis:
The North American region dominates the market with the largest market share due to the high prevalence of Herpes infection in the region. The market size is expected to grow at a CAGR of 5.1% (2023-2032) against an earlier estimation of $1.6 billion in 2022 to grow a worth around $2.6 billion by 2032.
The European market is expected to grow at a significant rate due to the rising incidence of Herpes infection and increasing awareness about the disease. The market size is expected to grow at a CAGR of 4.8% (2023-2032) against an earlier estimation of $1.2 billion in 2022 to grow a worth around $2.0 billion by 2032.
The Asia Pacific region is expected to witness significant growth in the Herpes infection treatment market due to the high prevalence of the disease and increasing healthcare infrastructure. The market size is expected to grow at a CAGR of 6.2% (2023-2032) against an earlier estimation of $1.0 billion in 2022 to grow worth around $1.8 billion by 2032.
The Latin American market is expected to witness moderate growth due to the increasing prevalence of the disease and rising healthcare expenditure. The market size is expected to grow at a CAGR of 4.5% (2023-2032) against an earlier estimation of $0.5 billion in 2022 to grow a worth around $0.8 billion by 2032.
The Middle East and African market are expected to witness slow growth due to low healthcare expenditure and a lack of awareness about the disease. The market size is expected to grow at a CAGR of 3.8% (2023-2032) against an earlier estimation of $0.2 billion in 2022 to grow a worth around $0.3 billion by 2032.
Herpes Infection Treatment Challenges:
The Herpes infection treatment market faces several challenges, including:
- Lack of awareness and funds: Some people could be reluctant to hunt for assistance or treatment because they lack awareness of Herpes, its symptoms, and its transmission as well as funds for treatment.
- Lack of vaccines: There's no vaccine for the Herpes simplex virus at this point, although some vaccines are in development and a few clinical trials are still ongoing.
- Limited efficacy of current drugs: Although current medications for Herpes can alleviate symptoms, they are not eradicating the disease, and thus the virus can still spread.
- Reimbursement issues: In some nations, reimbursement coverage for Herpes treatment is restricted, making treatment less accessible to some individuals.
- Resistance to antiviral drugs: Herpes simplex virus can develop resistance to antiviral medications over time, which may make treatment less effective.
- Side effects of treatment: Side effects of antiviral medications used to treat Herpes can include nausea, headache, and diarrhea.
- Stigma and discrimination: Herpes patients could be subjected to discrimination and stigma, which can prevent them from seeking treatment and sharing their condition with others.
Herpes Infection Treatment Market Key Players:
There are several key players in the Herpes Infection Treatment market, including:
- Astellas Pharma Inc: This is a Japanese multinational pharmaceutical company that markets acyclovir, valacyclovir, and famciclovir.
- Gilead Sciences, Inc: This is an American biopharmaceutical company that markets acyclovir, valacyclovir, and famciclovir. It produces a medication called Valtrex (valacyclovir), which is used to treat genital Herpes, Herpes zoster (shingles), and cold sores.
- GlaxoSmithKline plc: This British multinational pharmaceutical company sells other Herpes medications like acyclovir, valacyclovir, and famciclovir. GlaxoSmithKline produces a medication called Zovirax (acyclovir), which is used to treat genital Herpes, Herpes zoster (shingles), and chickenpox.
- Merck & Co., Inc: Merck & Co. produces a medication called ProQuad, which is a combination vaccine used to protect against measles, mumps, rubella, and varicella (chickenpox) also markets acyclovir, valacyclovir, and famciclovir.
- Mylan N.V.: Mylan N.V. produces a medication called Valacyclovir Hydrochloride Tablets, which is used to treat genital Herpes, Herpes zoster (shingles), and cold sores.
- Novartis International AG: Novartis International AG produces a medication called Famvir (famciclovir), which is used to treat Herpes zoster (shingles) and genital Herpes.
- Pfizer Inc.: This is an American multinational pharmaceutical corporation that markets acyclovir, valacyclovir, and famciclovir.
- Sun Pharmaceutical Industries Ltd.: Sun Pharmaceutical Industries produces a medication called Acivir, which is used to treat Herpes simplex virus (HSV) infections, including genital Herpes and cold sores.
- Teva Pharmaceutical Industries Ltd.: Teva Pharmaceutical Industries produces a medication called Acyclovir, Valacyclovir, and Famciclovir which is used to treat Herpes simplex virus (HSV) infections, including genital Herpes and cold sores.
- Viatris Inc.: This is an American global pharmaceutical company that markets acyclovir, valacyclovir, and famciclovir.
|
Herpes Infection Treatment Market: Report Scope |
|
|
Base Year |
2022 |
|
Base Year Market Size |
$7.0 billion |
|
Forecast Year |
2023-2032 |
|
Forecast Year Market Size |
$11.2 billion |
|
CAGR Value |
4.8% |
|
Segmentation |
|
|
Challenges |
|
|
Growth Drivers |
|

Need Customized Report for Your Business ?
Utilize the Power of Customized Research Aligned with Your Business Goals
Request for Customized Report- Quick Contact -
- ISO Certified Logo -